Nanoparticle Characterization and Surface Chemistry
|
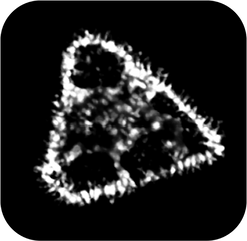
Whether they will be used in catalysis or artificial limbs, nanoparticle surfaces influence every aspect of their behavior. The ligand shell of a nanocrystal can determine its luminescence, its performance in a solar cell, or its clearance from the human body – to name just a few examples. We are interested in developing rapid, readily accessible methods to characterize and control this nanoparticle surface architecture – both the crystallographic and chemical composition – in order to develop new nanoparticle morphologies and reaction mechanisms that will have applications in fields ranging from catalysis to medicine.
Active Collaborator: Prof. Sunil Saxena, Pitt Chemistry
|
Multifunctional Nanoparticle Synthesis
|
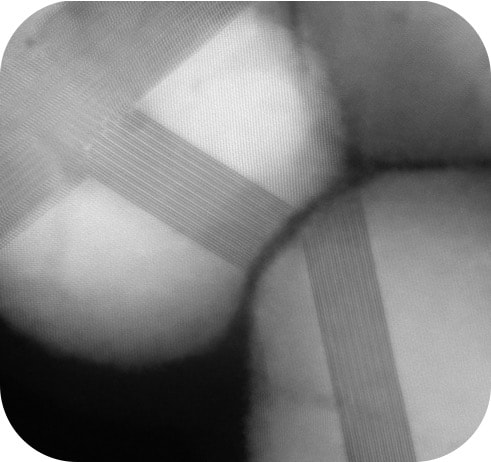
It is well known that the physical properties of nanoscale materials are highly dependent on their morphology (size, shape, surface chemistry, composition, and composition architecture). These relationships imply that for every nanoparticle composition (and combination of compositions), there may be an infinite number of optical, electronic, and catalytic behaviors. However, while the variety of particle possibilities may be large, there is currently no systematic way to design and then rationally access a particular nanoparticle architecture.
Elucidating these pathways would allow us to better use our current materials, and more effectively tailor new ones. These innovations require advances in both fundamental chemistry knowledge and the analytical methods used to track them. Just as organic chemistry research has developed a mechanistic framework and synthetic toolkit that has produced everything from plastics to pharmaceuticals, so too must these concepts be developed for nanochemistry in order to harness the similar potential of nanomaterials. Through the discovery and tracking of nanoparticle reaction mechanisms, we work to develop a set of physical, analytical, and synthetic principles to rationally generate complex, highly-tailored nanoparticles for environmental remediation and catalysis applications. Active Collaborators: Prof. Nathaniel Rosi, Pitt Chemistry; Prof. Renee Frontiera, University of Minnesota Chemistry; Dr. Sadegh Yazdi, University of Colorado Boulder
|
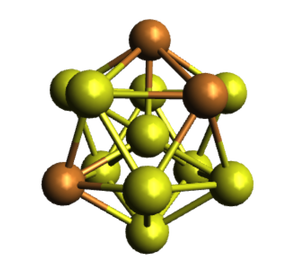
"Small" Nanoparticle Alloys
|
Small, multimetallic nanoparticles (diameter ~ 2-5 nm) promise to provide improved catalysts for efficient use of fossil fuel resources as well as multifunctional tools in biomedical applications. They also exist at an exciting interface between molecular and metallic electronic structure in nanomaterials. At this length scale, we study the fundamental chemical forces controlling nanoparticle composition, atomic structure, and optoelectronic properties.
Active Collaborators: Prof. John Kitchin, Carnegie Mellon University Chemical Engineering; Prof. Stefan Bernhard, Carnegie Mellon University Chemistry; Prof. Zachary Ulissi, Carnegie Mellon University Chemical Engineering
|
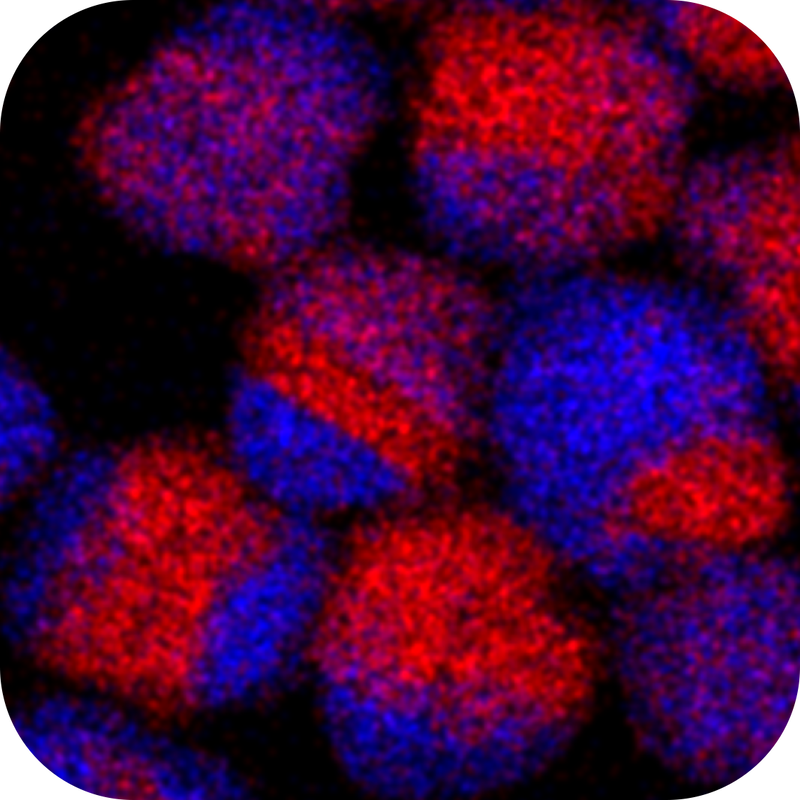
Nano-Bio Interactions
|
When implemented in technologies, nanomaterials interact with humans and the environment - whether by consequence or by design. We collaborate with engineers and physicians to understand the properties of nanomaterials that drive those interactions. Guided by discoveries in these collaborations, we use our expertise in synthesis and surface chemistry to develop nanomaterials with predictable and desirable mechanisms of response at the interface between nanomaterials and the broader world.
Active Collaborators: Prof. Leanne Gilbertson, Pitt Civil and Environmental Engineering
|

The Millstone Group Website by Jill Millstone is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Based on work at www.pitt.edu/~jem210 and websites therein.
**The creative commons license means you're welcome to use our text as long as you attribute it to our website. For example, when using our website research explanations (or some slightly modified version of these) in journal articles or other websites.**